ABSTRACT
Wearable sensors are susceptible to degradation from physical wear, moisture, and desiccation, which can result in signal attenuation and unreliable data. This pilot study, conducted in a controlled single-participant setting, introduces a framework to quantify and characterize sensor degradation while restoring corrupted electromyography (EMG) signals. Four types of sensors—polyethylene terephthalate film, parylene film, 3M bioelectrode pads, and microneedle patches—were affixed to the left forearm in a three-electrode EMG configuration. Impedance at 100 Hz was monitored as an indicator of sensor aging, while a one-dimensional convolutional autoencoder was employed to reconstruct degraded EMG signals using a loss function that incorporated both time-domain and frequency-domain error terms. The reconstruction loss showed a correlation with impedance changes, providing a practical metric for assessing sensor health. These findings highlight the feasibility of real-time signal recovery and its potential to extend the lifespan of sensors.
-
KEYWORDS: Wearable sensor, Signal restoration, Self-assessment, Performance degradation
-
KEYWORDS: 웨어러블 센서, 신호 복원, 자가진단, 성능 저하
1. Background
Wearable sensors are increasingly used in health monitoring, fitness tracking and environmental sensing. However, routine use exposes them to mechanical abrasion, desiccation, and perspiration, which accelerate performance degradation [
1-
3]. Such degradation reduces measurement accuracy and attenuates signals, potentially causing misinterpretation of physiological state and undermining clinical decision-making [
4,
5]. Recent advances in epidermal and stretchable EMG electrodes have enabled conformal and motion-tolerant recording, yet these studies primarily focus on sensor design or short-term performance, rather than quantitatively tracking progressive degradation or restoring signals [
6,
7]. To mitigate these risks, we present a technique that simultaneously assesses sensor integrity and corrects signal distortion, thereby preserving data fidelity and extending device lifetime. Four representative electrodes were evaluated: a rigid polyethylene terephthalate (PET) OHP film, an ultrathin parylene film, a 3M commercial hydrogel pad, and a microneedle (MN) patch [
6]. Electromyography (EMG) was recorded in a three-electrode configuration, with working and counter electrodes on the inner forearm and a reference electrode on the wrist. Sensor aging was quantified by measuring skin-electrode impedance at 100 Hz, within the 20-500 Hz EMG band yet outside power-line harmonics [
7]. Changes in signal-to-noise ratio (SNR) and spectral content were analyzed using visualization and Fast Fourier Transform (FFT). Unlike prior studies, this framework jointly tracks impedance, SNR, and spectral features under degradation across multiple wearable EMG sensors, while restoring degraded signals and providing a self-assessment metric for sensor health. This framework is designed to support reliable, long-term deployment of wearable sensor technology in both clinical and daily-life applications.
2. Research Objectives
This study aims to improve the reliability and lifetime of wearable EMG systems under degradation conditions, particularly when prolonged recording is demanded [
8]. For this purpose, four specific goals were defined. The first objective is to quantify the performance degradation of four representative wearable EMG sensors—PET OHP film, 3M commercial bioelectrode pads, parylene film, and MN patches—by tracking impedance at 100 Hz throughout the degradation process [
9]. Building on this analysis, the second objective is to characterize the distribution of SNR and degradation patterns across sensors, enabling investigation of sensor-specific aging mechanisms and evaluation of signal restoration feasibility. Third, a one-dimensional convolutional autoencoder model was employed to reconstruct degraded EMG signals [
10,
11]. Finally, the model’s reconstruction loss was used as a lightweight, on-device metric for self-assessment of sensor condition [
12-
14]. This pilot study is confined to laboratory benchtop aging experiments and signal restoration. Future work will involve real-time embedded deployment and multi-participant validation.
3. Materials & Methods
Fig. 1 illustrates the four representative EMG electrode types prepared for this study. The MN patch is a rigid polymer and is composed of 10 by 10 array of conical needles, each 850 μm in height. The entire surface was coated with a 100 nm-thick gold layer to ensure low-impedance signal acquisition [
15]. A 600 nm-thick parylene-C membrane, coated with 100 nm of gold, was fabricated to yield a flexible, skin-conformal electrode [
16]. A 100 μm-thick commercial PET OHP film was cut and coated with gold, producing a rigid electrode. 3M commercial hydrogel bioelectrode pads were used without modification and served as both test electrodes (as working and counter positions) and as the constant reference electrode throughout all trials.
After fabrication and preparation, a three-electrode layout was adopted. The working and counter electrodes were affixed to the ventral forearm, 5 cm apart for optimal measurement, while the reference electrode was placed on the outer wrist [
17]. Each sensor type was tested sequentially on the same location, with the surface cleansed with phosphate-buffered saline and water, followed by rest time to minimize variability.
Electrodes were connected to an EMG signal acquisition breadboard. The output was monitored using a Yokogawa DL1540C oscilloscope and digitized via an Arduino Leonardo with a sampling rate of 1 kHz. Data were streamed via Arduino IDE and processed using a Python script. From each 40-second recording, the central 30 seconds were extracted for analysis to avoid motion artifacts. The subject was instructed to maintain a constant voluntary contraction to produce repeatable EMG bursts of moderate amplitude.
To emulate realistic sensor degradation, each sensor was exposed to a combination of mechanical and environmental stressors, including desiccation, bending, rear-side abrasion, PBS infusion, and mild heating. Sensor aging is quantified with the impedance measurement at the skin-electrode interface. For each degradation level, a corresponding EMG signal was recorded. Since most surface EMG signal energy lies within the 10-250 Hz range, and amplifier bandwidths are typically specified at 10-500 Hz, a minimum sampling rate of 1 kHz was used to prevent information loss in accordance with the Nyquist criterion [
18]. Skin-electrode impedance was recorded at 100 Hz using a Keysight U1773C LCR meter, as this frequency lies within the EMG passband while avoiding power-line harmonics [
5].
Raw EMG traces were segmented into 1-second windows, zero-mean normalized, and input into a one-dimensional convolutional autoencoder (1-D CAE) developed using Keras. The model was trained to reconstruct 1-second EMG segments using a composite loss function combining time-domain mean squared error and frequency-domain spectral error. The architecture is summarized in
Table 1. The network was trained on segmented signals from the first fifteen high-quality MN recordings and subsequently applied to severely degraded samples for signal restoration.
4. Results
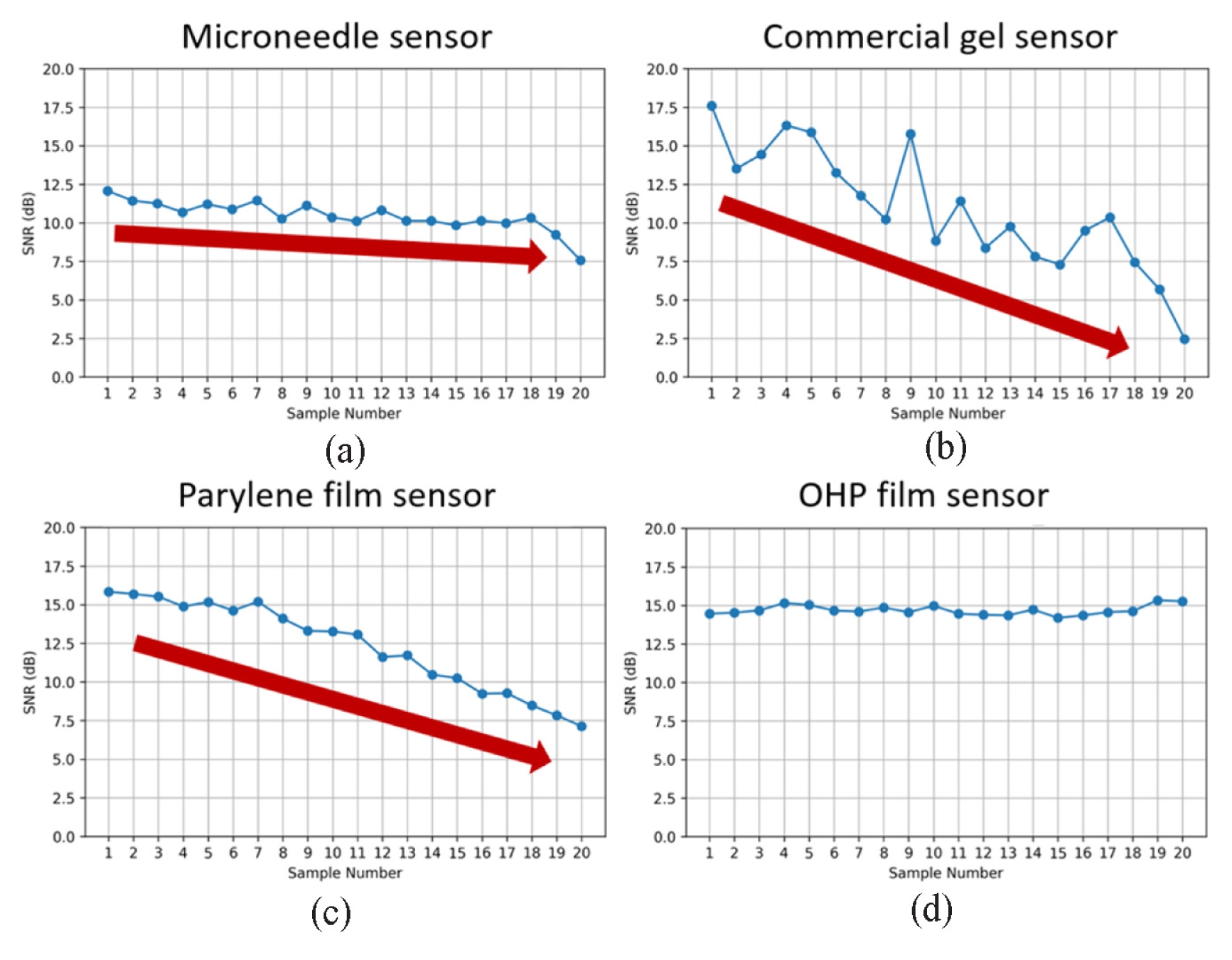
As shown in
Fig. 3, SNR is plotted against sample number, with samples arranged in order of increasing impedance for each sensor group. The impedance ranges (in kΩ) for the MN, commercial gel, parylene film, and PET OHP film electrodes were 2.64-73.92, 46.04-209, 36.71-156.23, and 19.72-74.08, respectively. The MN, commercial gel, and parylene sensors exhibited a clear monotonic decrease in SNR with increasing impedance. In contrast, the PET OHP electrode maintained a nearly constant SNR despite progressive impedance changes, indicating a weak correlation between interface degradation and signal quality.
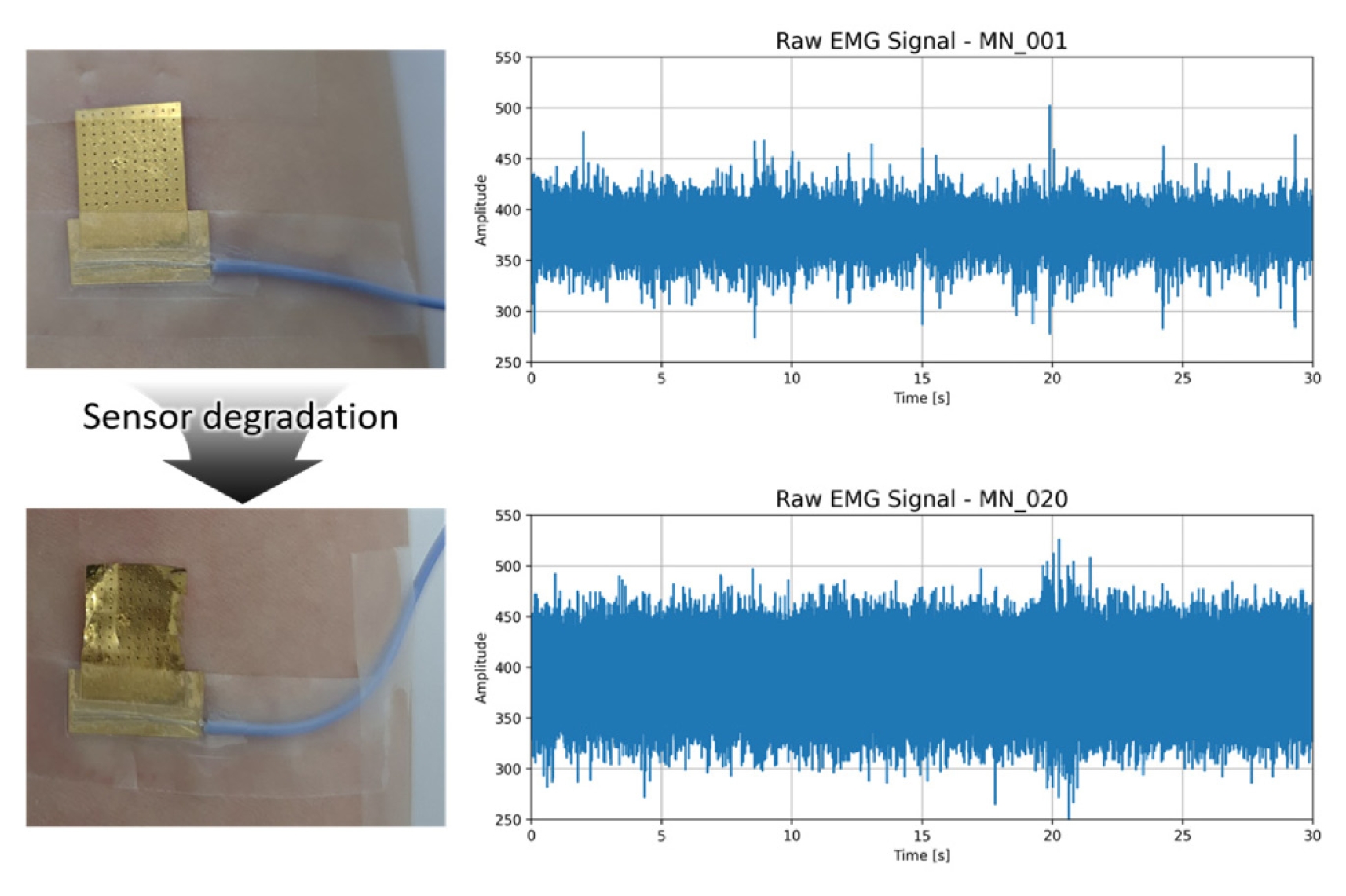
Fig. 4 compares the MN patch in its pristine state (Sample 1) with a severely degraded state (Sample 20). Visual inspection reveals delamination of the gold layer, surface cracks, and uneven surface. The corresponding raw EMG trace shows a rise in baseline noise, occasional dropout spikes and a reduced peak-to-peak muscle activity.
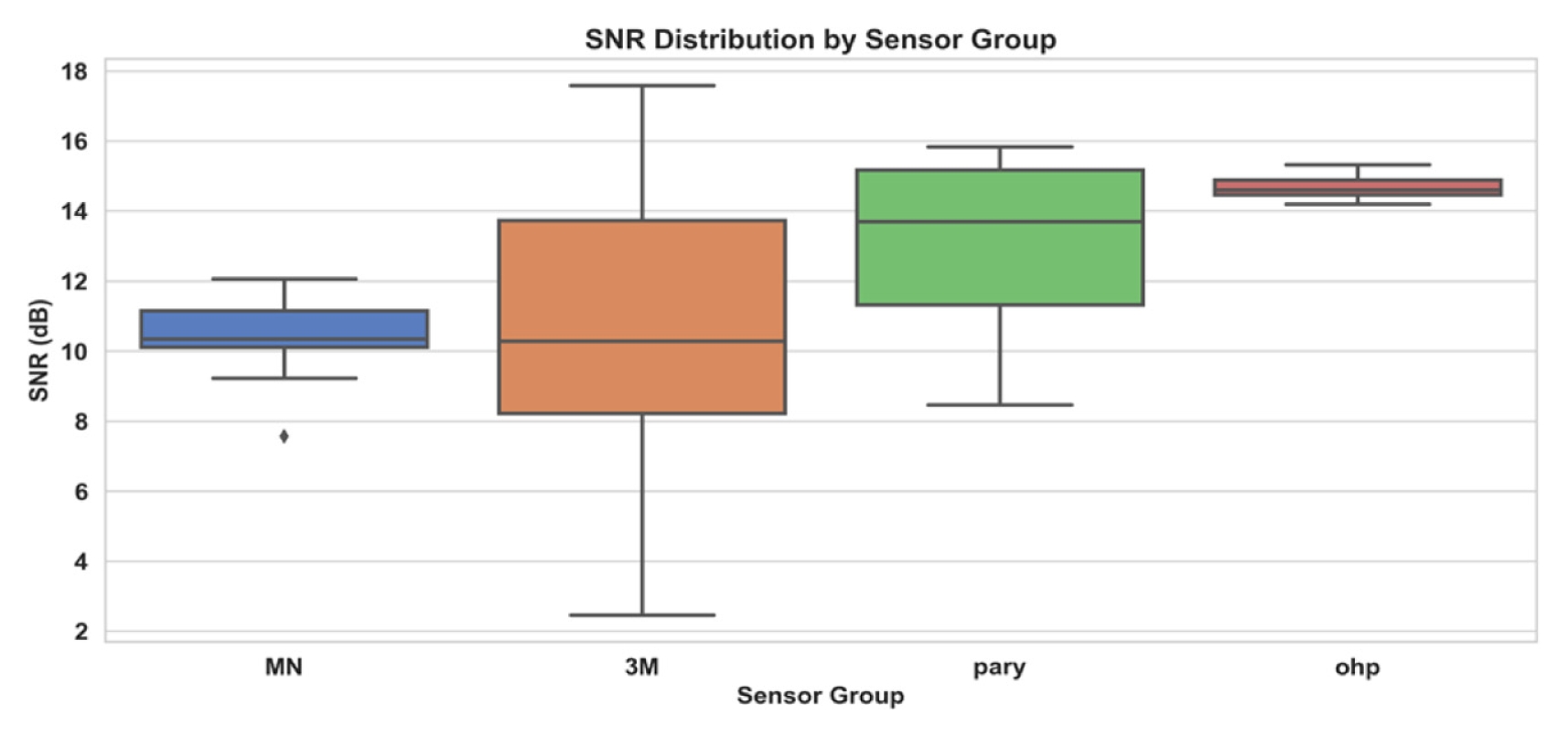
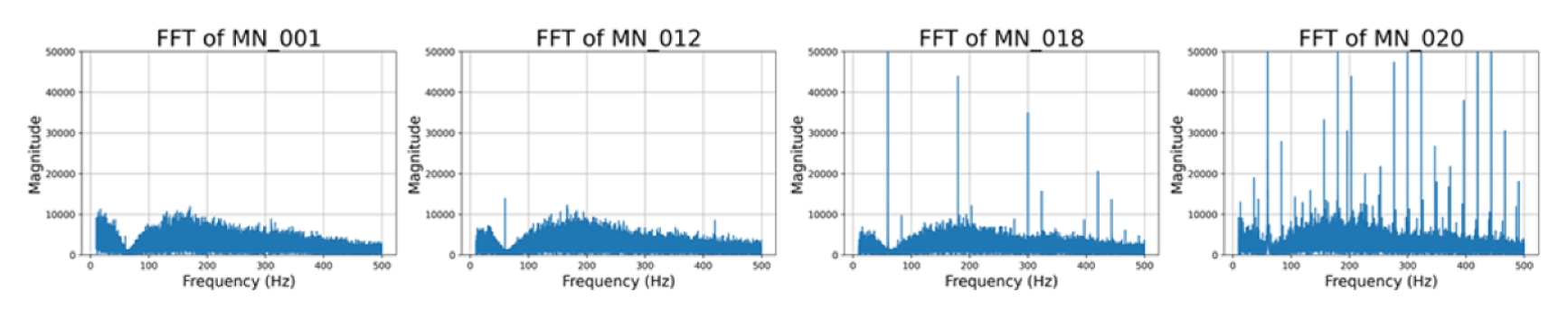
Fig. 5 presents a boxplot summarizing 20 recordings for each sensor type. Median SNR values were MN = 10 dB, commercial gel = 10 dB with the widest interquartile range, parylene = 14 dB, and PET OHP = 15 dB. The FFT spectra of four MN samples in
Fig. 6 illustrate a progressive increase in broadband noise and the emergence of narrow harmonics with sensor aging.
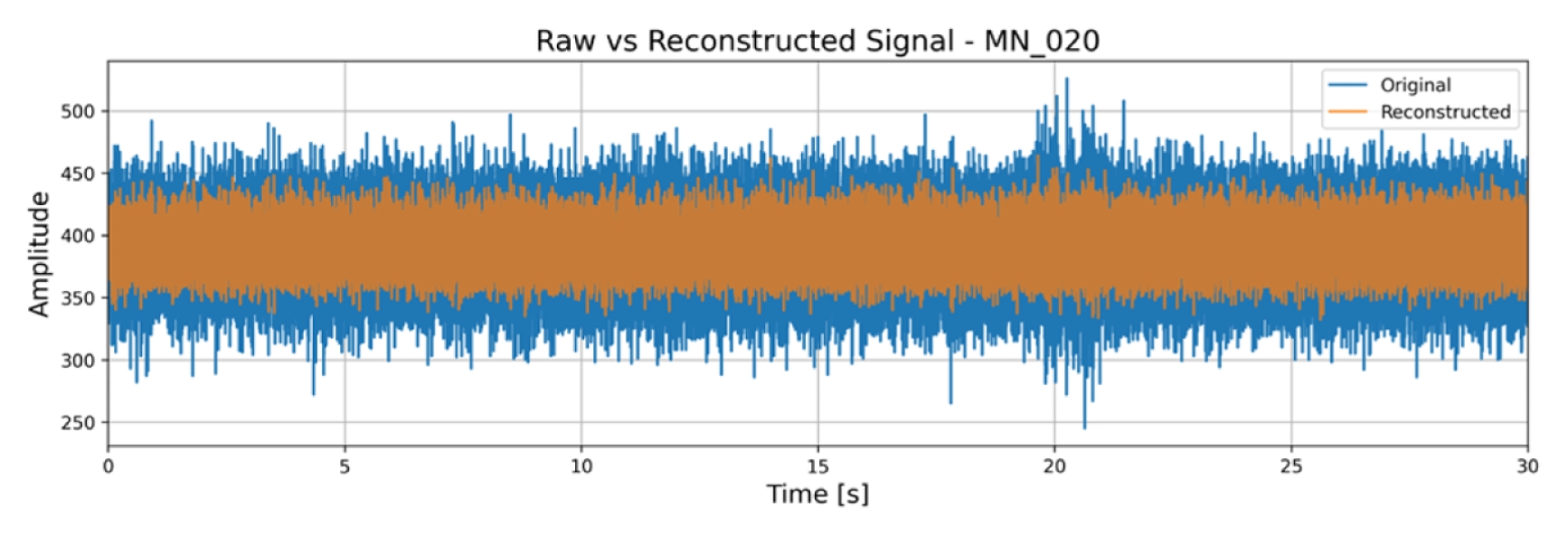
Fig. 7 overlays the raw and reconstructed EMG signals for a degraded MN sample. Baseline noise is suppressed, while the peak-to-peak amplitude of muscle bursts is preserved.
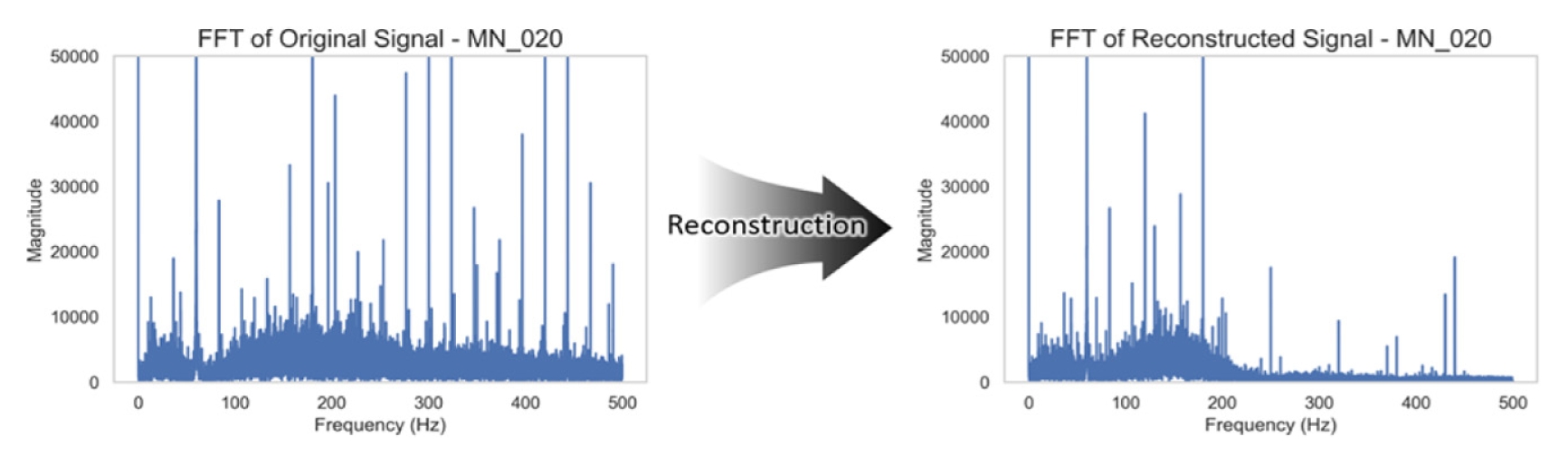
Fig. 8 summarizes the corresponding frequency-domain effects. The model successfully attenuated sharp harmonics introduced during sensor degradation while retaining the primary content of the EMG signal.
5. Discussion
The result reveals a strong relationship between skin-electrode impedance at 100 Hz and EMG signal quality. A strong negative correlation between impedance and SNR for the MN, commercial gel, and parylene electrodes confirms impedance as a reliable, sensor agnostic metric of degradation. In contrast, the absence of correlation in the rigid OHP electrodes suggests substrate stiffness can mask electrical deterioration, rendering impedance tracking invalid for degradation monitoring in such devices. Beyond this overall trend, failure modes varied by sensor type. Hydrogel desiccation dominated the degradation process in commercial gel pads, causing rapid impedance increases and a wide spread in SNR values [
19]. In parylene films, mechanical wear led to progressive degradation, resulting in a steady decline in SNR. MN patches benefited from partial tissue penetration, which helped stabilize the contact and delayed catastrophic failure [
15]. Notably, the flexible parylene membrane maintained the highest median SNR, suggesting that conformal contact mitigates noises until severe surface wear occurs. The 1-D CAE restored SNR and suppressed baseline noise without introducing spectral distortion. Because the model tracks reconstruction loss during forward propagation, it enables a lightweight algorithm to both assess and correct signal degradation.
6. Conclusion
Real-time impedance monitoring, coupled with embedded denoising via 1-D CAE, can extend the useful lifetime of wearable sensors, reduce recalibration frequency, and lower both consumable costs and clinic visits in long-term applications such as prosthetic control or rehabilitation feedback. This pilot study involved a single participant, and larger cohorts are required to confirm the generalizability. Further analysis of degradation patterns in relation to film material properties and thickness will be addressed in future work. These findings can guide the design of durable wearable EMG systems for clinical rehabilitation and long-term home monitoring. Moreover, the proposed framework has potential applications in biosensing fields, such as extending attachment times and improving signal stability in EMG-based muscle fatigue studies, and reducing EMG signal contamination and solving wet-electrode issues in brain-computer interfaces studies, ultimately enhancing the reliability of long-duration recordings [
20,
21].
FOOTNOTES
-
ACKNOWLEDGEMENT
This study was supported by a faculty research grant of Yonsei University College of Medicine (No. 6-2023-0087) and by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (No. RS-2024-00452785).
Fig. 1Four wearable EMG sensors. (a) Microneedle patch comprising a 10 × 10 array of 850 μm tall needles, (b) 3 M commercial monitoring electrode, (c) 600 nm thick flexible parylene-C film, and (d) 100 μm thick rigid PET OHP film electrode

Fig. 2Experimental setup for degradation testing and EMG acquisition. (a) Three-electrode configuration: working and counter electrodes placed 5 cm apart on the forearm, reference electrode on the wrist, (b) Demonstration of intentional sensor degradation process

Fig. 3SNR versus sample number for (a) microneedle, (b) commercial gel, (c) parylene film and (d) PET OHP film sensors. Red arrows indicate overall decaying trend
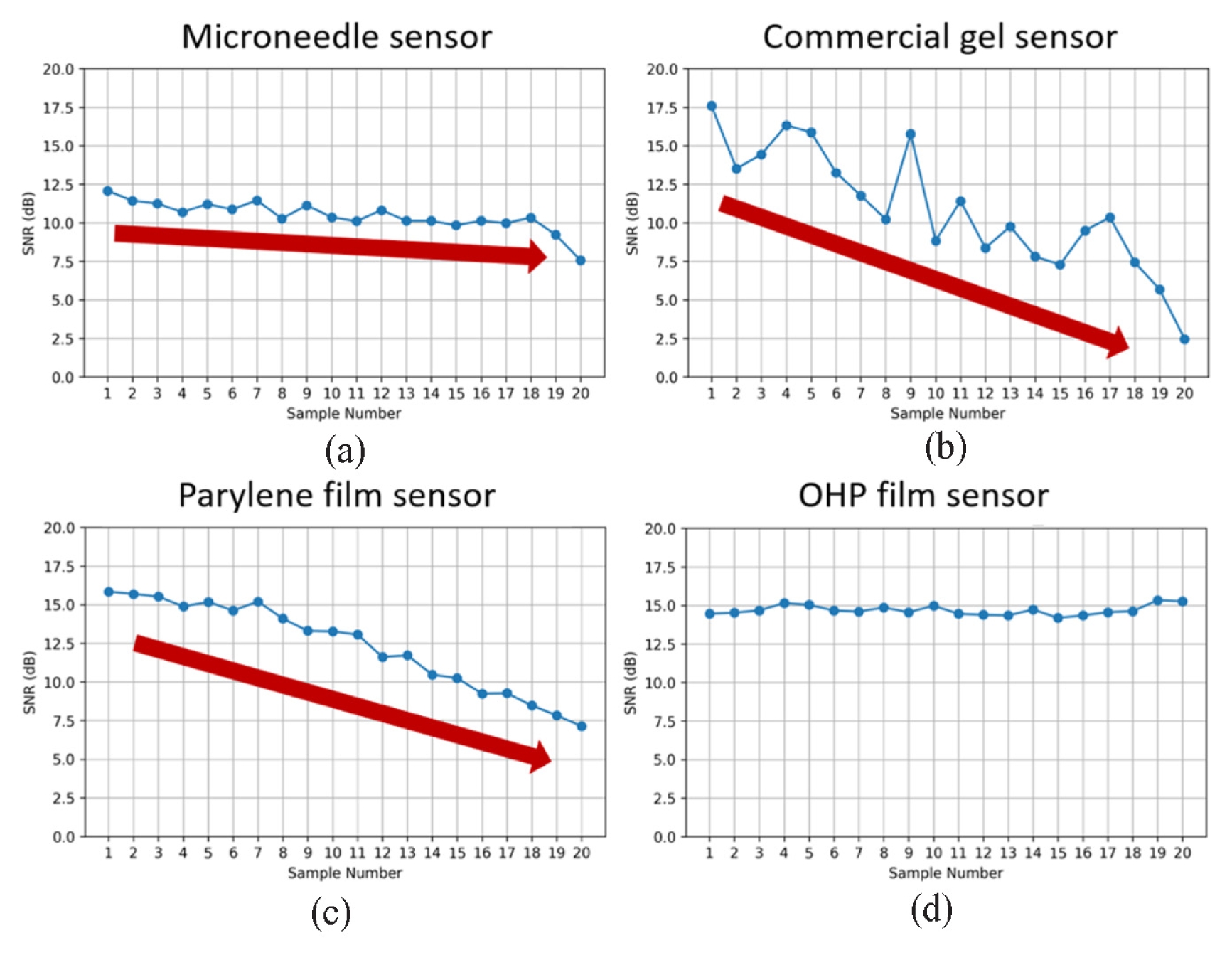
Fig. 4Microneedle sensor before (top) and after (bottom) degradation, paired with 30 seconds raw EMG signals. Note surface deformation and elevated baseline noise
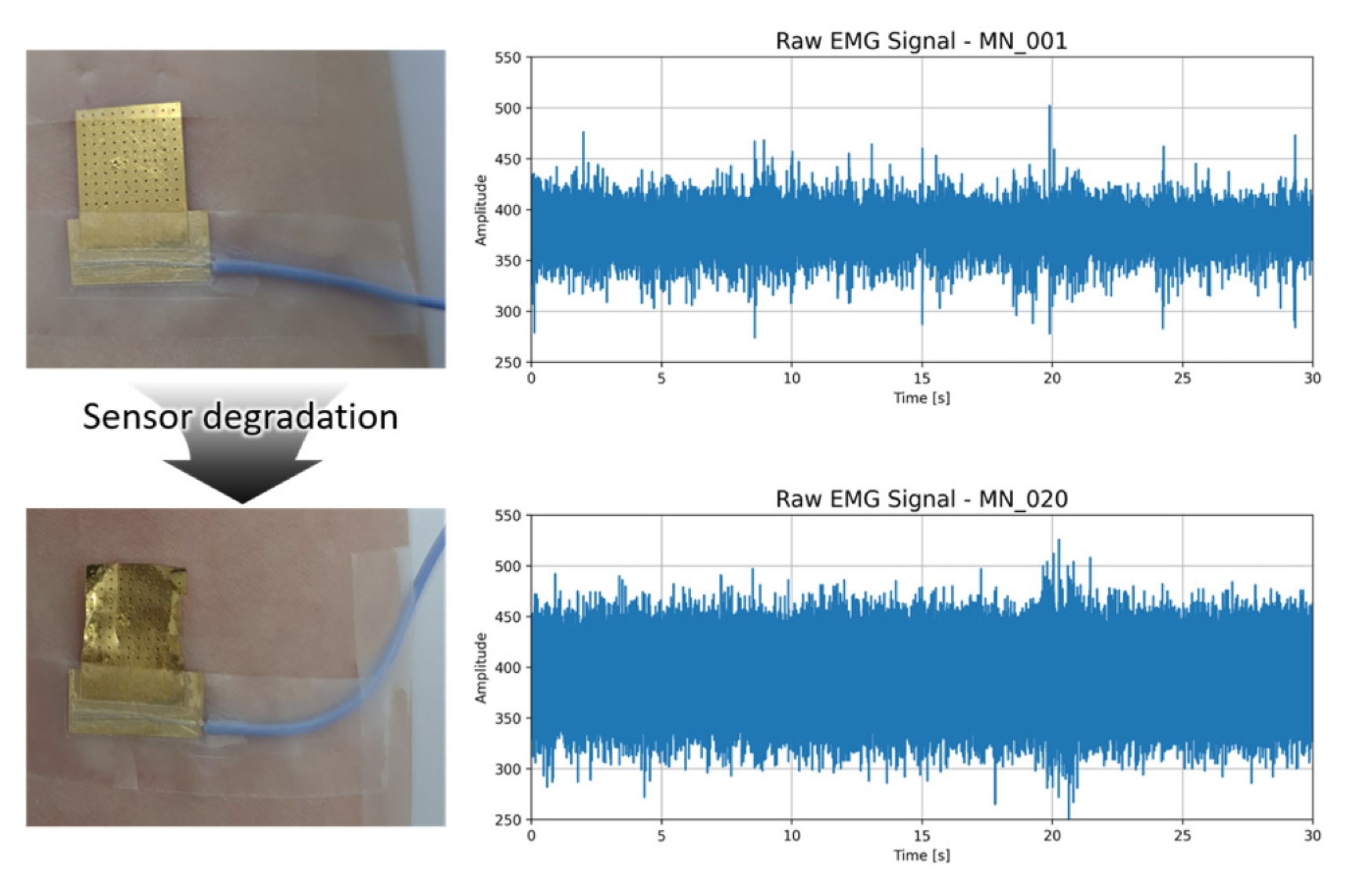
Fig. 5Boxplot of SNR distribution by sensor groups (n = 20)
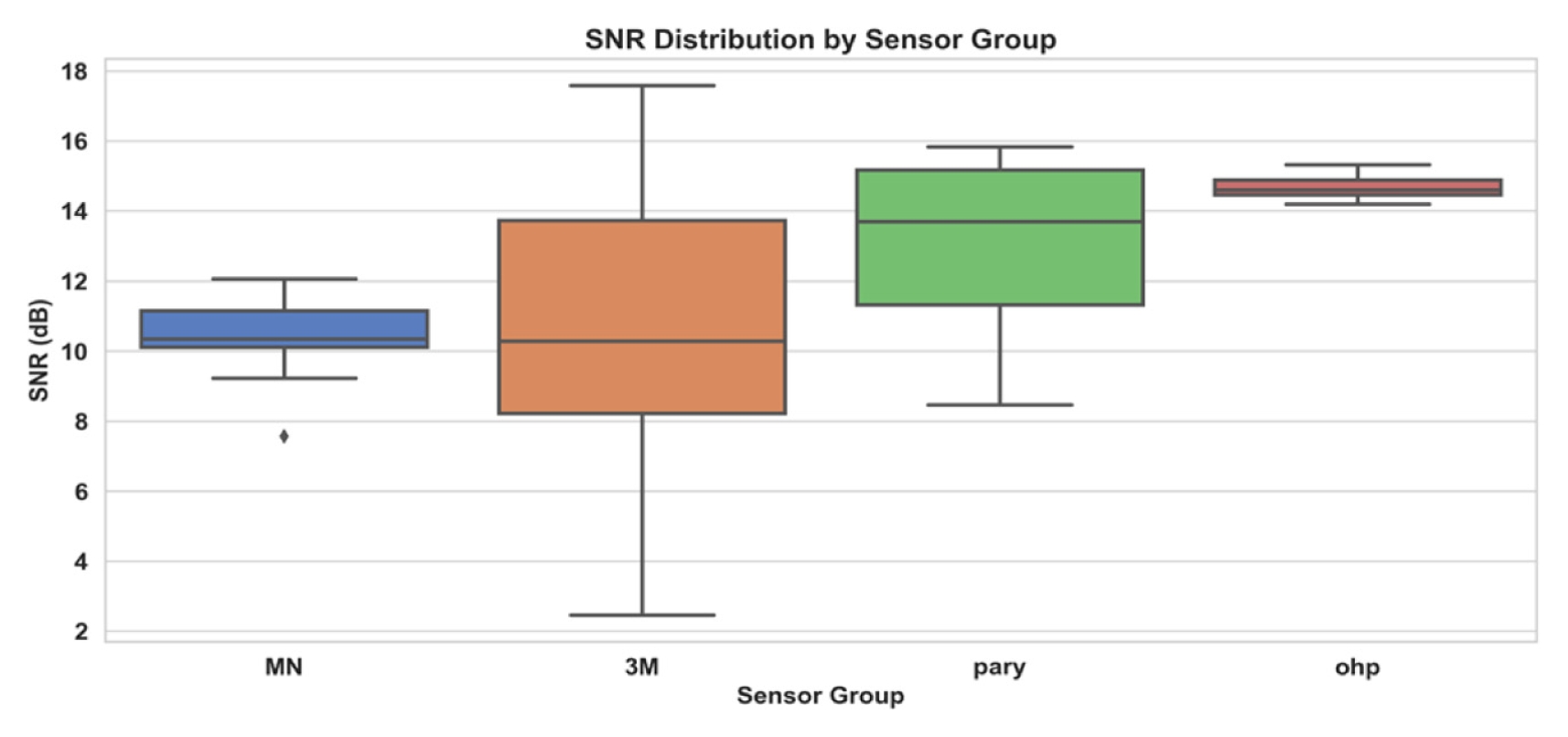
Fig. 6FFT magnitude spectra of MN recordings, illustrating gradual noise growth and harmonic spikes
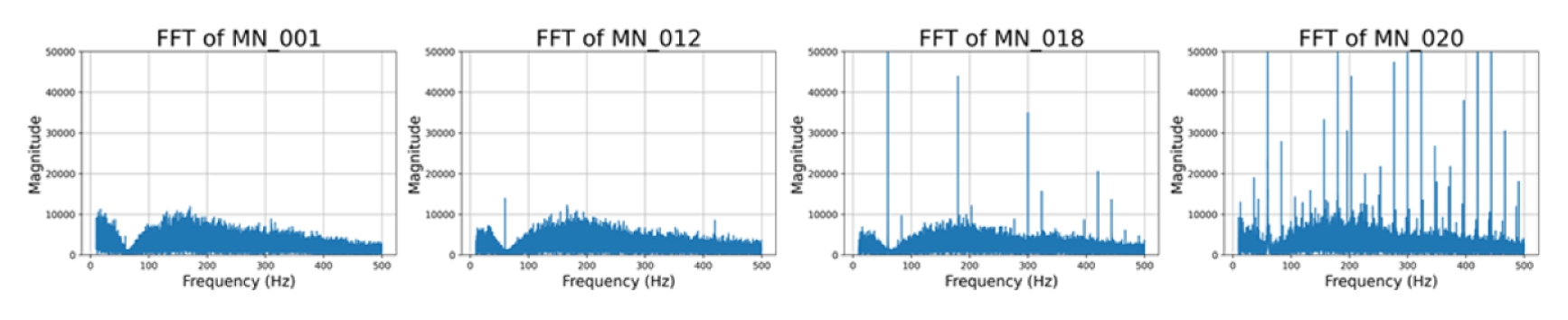
Fig. 7Time-domain overlay of raw (blue) and autoencoder reconstructed (orange) EMG for degraded MN EMG signal of sample 20. Denoising visibly suppresses baseline noise
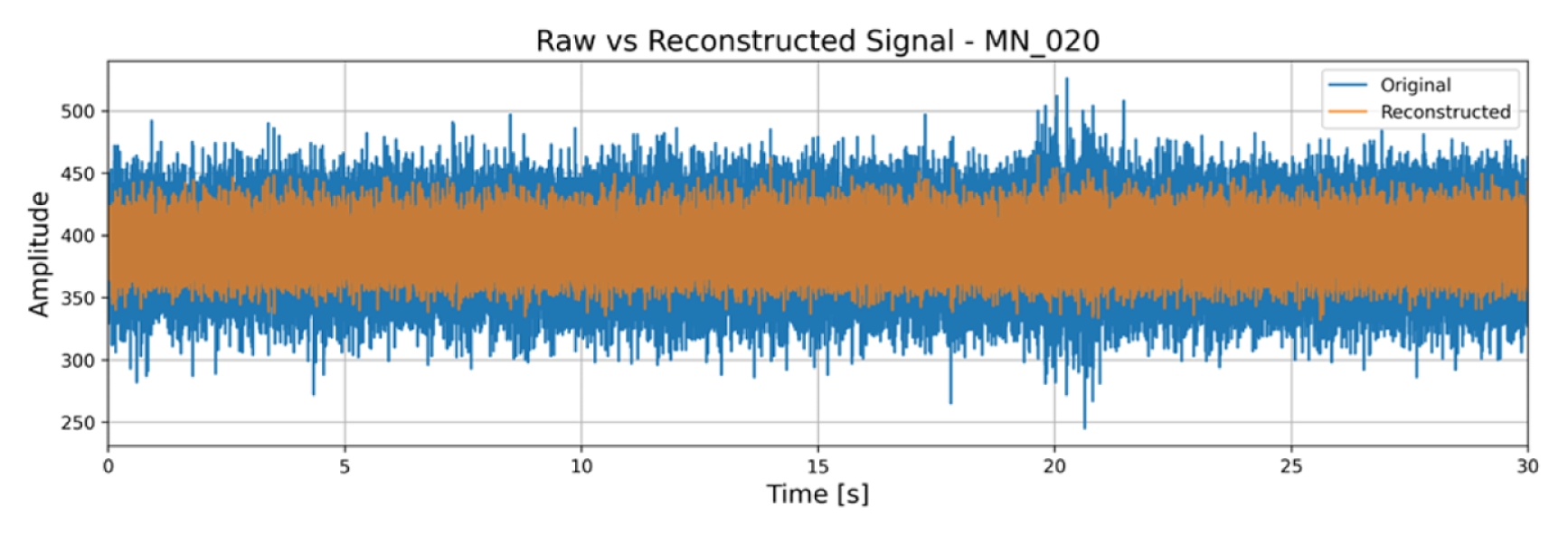
Fig. 8FFT comparison of the same signal pair. Reconstruction attenuates harmonic noise peaks, while preserving low-frequency muscle energy
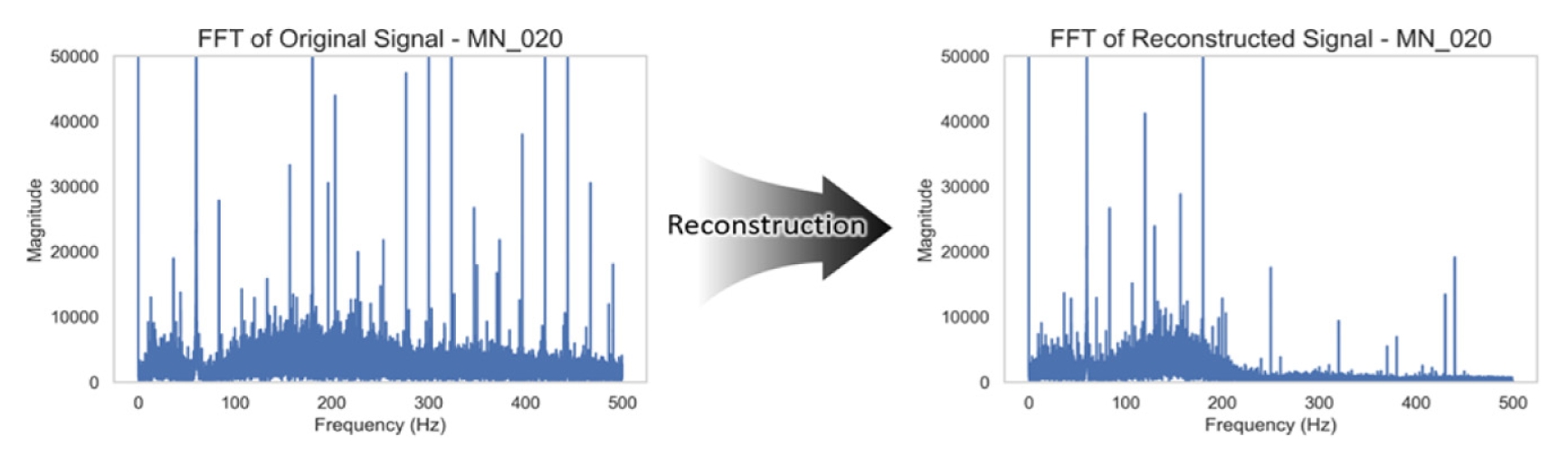
Table 1The Structure of the 1-D convolutional autoencoder
Table 1
|
Stage |
Layer |
Output shape |
|
Input |
|
1,000 × 1 |
|
Encoder |
Conv1D (16 filters, kernel = 3, ReLU) |
1,000 × 16 |
|
MaxPooling1D |
500 × 16 |
|
Conv1D (8 filters, kernel = 3, ReLU) |
500 × 8 |
|
MaxPooling1D |
250 × 8 |
|
Decoder |
Conv1D (8 filters, kernel =3, ReLU) |
250 × 8 |
|
UpSampling1D |
500 × 8 |
|
Conv1D (16 filters, kernel = 3, ReLU) |
500 × 16 |
|
UpSampling1D |
1,000 × 16 |
|
Output |
Conv1D (1 filter, kernel = 3, sigmoid) |
1,000 × 1 |
REFERENCES
- 1. Ates, H. C., Nguyen, P. Q., Gonzalez-Macia, L., Morales-Narváez, E., Güder, F., Collins, J. J., Dincer, C., (2022), End-to-end design of wearable sensors, Nature Reviews Materials, 7(11), 887-907.
- 2. Heikenfeld, J., Jajack, A., Rogers, J., Gutruf, P., Tian, L., Pan, T., Li, R., Khine, M., Kim, J., Wang, J., Kim, J., (2018), Wearable sensors: modalities, challenges, and prospects, Lab on a Chip, 18(2), 217-248.
- 3. Bariya, M., Nyein, H. Y. Y., Javey, A., (2018), Wearable sweat sensors, Nature Electronics, 1(3), 160-171.
- 4. van Melzen, R., Haveman, M. E., Schuurmann, R. C., van Amsterdam, K., El Moumni, M., Tabak, M., Struys, M. M., de Vries, J-P. P, (2024), Validity and reliability of wearable sensors for continuous postoperative vital signs monitoring in patients recovering from trauma surgery, Sensors, 24(19), 6379.
- 5. McManus, L., De Vito, G., Lowery, M. M., (2020), Analysis and biophysics of surface EMG for physiotherapists and kinesiologists: Toward a common language with rehabilitation engineers, Frontiers in Neurology, 11, 576729.
- 6. Kim, D.-H., Lu, N., Ma, R., Kim, Y-S, Kim, R-H, Wang, S, Wu, J, Won, S. M., Tao, H., Islam, A., (2011), Epidermal electronics, Science, 333(6044), 838-843.
- 7. Yang, S., Cheng, J., Shang, J., Hang, C., Qi, J., Zhong, L., Rao, Q., He, L., Liu, C., Ding, L., (2023), Stretchable surface electromyography electrode array patch for tendon location and muscle injury prevention, Nature Communications, 14(1), 6494.
- 8. Searle, A., Kirkup, L., (2000), A direct comparison of wet, dry and insulating bioelectric recording electrodes, Physiological Measurement, 21(2), 271-283.
- 9. Goyal, K., Borkholder, D. A., Day, S. W., (2022), Dependence of skin-electrode contact impedance on material and skin hydration, Sensors, 22(21), 8510.
- 10. Zou, Y., Cheng, L., Han, L., (2023), Reconstruction of incomplete surface electromyography based on an adversarial autoencoder network, Biomedical Signal Processing and Control, 86, 105084.
- 11. Dinashi, K., Ameri, A., Akhaee, M. A., Englehart, K., Scheme, E., (2022), Compression of EMG signals using deep convolutional autoencoders, IEEE Journal of Biomedical and Health Informatics, 26(7), 2888-2897.
- 12. Vincent, P., Larochelle, H., Lajoie, I., Bengio, Y., Manzagol, P.-A., Bottou, L., (2010), Stacked denoising autoencoders: Learning useful representations in a deep network with a local denoising criterion, Journal of Machine Learning Research, 11(110), 3371-3408.
- 13. Malhotra, P., Ramakrishnan, A., Anand, G., Vig, L., Agarwal, P., Shroff, G., (2016), Lstm-based encoder-decoder for multi-sensor anomaly detection, arXiv preprint arXiv:1607.00148.
- 14. Wu, D., Chen, D., Yu, G., (2024), New health indicator construction and fault detection network for rolling bearings via convolutional auto-encoder and contrast learning, Machines, 12(6), 362.
- 15. Tang, X., Dong, Y., Li, Q., Liu, Z., Yan, N., Li, Y., Liu, B., Jiang, L., Song, R., Wang, Y., (2023), Using microneedle array electrodes for non-invasive electrophysiological signal acquisition and sensory feedback evoking, Frontiers in Bioengineering and Biotechnology, 11, 1238210.
- 16. Peng, H.-L., Liu, J.-Q., Dong, Y.-Z., Yang, B., Chen, X., Yang, C.-S., (2016), Parylene-based flexible dry electrode for bioptential recording, Sensors and Actuators B: Chemical, 231, 1-11.
- 17. Hermens, H. J., Freriks, B., Disselhorst-Klug, C., Rau, G., (2000), Development of recommendations for SEMG sensors and sensor placement procedures, Journal of Electromyography and Kinesiology, 10(5), 361-374.
- 18. Konrad, P., (2005), The ABC of EMG: A practical introduction to kinesiological electromyography (Version 1.0). Noraxon U.S.A., Inc.
- 19. Sousa, A. S., Noites, A., Vilarinho, R., Santos, R., (2023), Long-term electrode–skin impedance variation for electromyographic measurements, Sensors, 23(20), 8582.
- 20. Nam, D., Park, K., (2022), Assessment of muscle fatigue using emg under long duration and high intensity fatigue condition, Journal of the Korean Society for Precision Engineering, 39(6), 433-441.
- 21. Lee, S., Jang, Y., Lee, H., Tae, K., (2024), Development of an EEG-based gait recognition classification CNN-BiLSTM model for brain-computer interfaces (BCI), Journal of the Korean Society for Precision Engineering, 41(6), 481-488.
Biography
- Juhyeong Jeon
Ph.D. candidate in the Department of Mechanical Engineering, POSTECH. His research interest is precision engineering.
- Gaeun Yun
Ph.D. candidate in the Department of Mechanical Engineering, POSTECH. Her research interest is precision engineering.
- Phuong Thao Le
Ph.D. candidate in the Division of Interdisciplinary Bioscience and Bioengineering, POSTECH. Her research interest is biotechnology and bioengineering.
- Jungho Lee
Affiliate Professor in the Department of Mechanical Engineering, POSTECH. His research interest is biomedical engineering.
- Tae Sik Hwang
Professor in the Department of Emergency Medicine, Sungkyunkwan University Samsung Changwon Hospital. His research interest is biotechnology.
- Geunbae Lim
Professor in the Department of Mechanical Engineering, POSTECH. His research interest is nano and bio- MEMS.
 , Tae Sik Hwang4,#
, Tae Sik Hwang4,# , Geunbae Lim1,2,#
, Geunbae Lim1,2,#