ABSTRACT
The field of tissue engineering requires versatile scaffold fabrication technologies capable of inducing cell proliferation and differentiation to promote functional tissue regeneration. Traditional fabrication methods face inherent trade-offs among production speed, resolution, and cost, which hinder their ability to replicate the intricate hierarchical structures of biological tissues. To address these challenges, we developed a mask projection photolithography system with variable optical magnification. This system allows for precise control of the microscale feature size in the final product using a single mask, by adjusting the optical magnification with interchangeable objective lenses and a relay lens. With this system, we successfully fabricated porous scaffolds with reproducible pore sizes ranging from 25 to 100 μm, exposing a Poly (ethylene glycol) diacrylate (PEGDA, Mn = 700) hydrogel precursor solution through a honeycomb-patterned mask for durations of just 3 to 10 seconds. The mask projection system presented in this study offers a powerful and efficient platform for creating the microstructures essential for various advanced biomedical applications, including tissue engineering, drug delivery, and organoid-on-a-chip, thanks to its unique combination of speed, precision, and cost-effectiveness.
-
KEYWORDS: Mask projection lithography, Hydrogel scaffold, Variable magnification, Pore size control, Tissue engineering
-
KEYWORDS: 마스크 투영 리소그래피, 하이드로겔 스캐폴드, 가변 배율, 기공 크기 제어, 조직 공학
NOMENCLATURE
Focal Length of the Relay/Collimation Lens
Focal Length of the Objective Lens
1. Introduction
Tissue engineering is an interdisciplinary field that utilizes three key elements such as cells, growth factors, and scaffolds to restore, maintain, or improve the function of damaged tissues or organs [
1]. Among these, the scaffold plays a crucial role beyond providing a mere physical support base; it provides a three-dimensional microenvironment that orchestrates a series of biological processes such as cell attachment, proliferation, and differentiation [
2,
3]. Therefore, fabricating scaffolds that can precisely mimic the complex hierarchical structure of natural tissues is a decisive factor in determining the success of tissue regeneration [
4]. Over the past few decades, various scaffold fabrication techniques have been developed, yet each has shown distinct limitations due to a fundamental trade-off among four key performance indicators: speed, resolution, flexibility, and cost [
5,
6]. Traditional techniques like salt leaching are inexpensive and simple but lack the precision to control pore size, shape, and interconnectivity, resulting in low reproducibility [
7]. Electrospinning can produce nanofibrous structures similar to the extracellular matrix (ECM), but the dense fiber network impedes deep cell penetration and nutrient transport [
8,
9].
Additive manufacturing technologies, such as 3D printing, have opened up possibilities for fabricating complex 3D structures with high precision based on computer-aided design (CAD) models [
10]. However, extrusion-based printing has limited resolution in the hundreds of micrometers and slow processing speeds. Conversely, projection technologies based on Digital Light Processing (DLP) using digital micromirror devices (DMDs) can achieve both high resolution at the micrometer scale and rapid fabrication speeds [
11-
13]. This technology, however, requires expensive DMD chips and precision optics, leading to high system setup costs. Furthermore, there is an inverse relationship between the resolution of the projected image and the total build area, which constrains the fabrication of fine patterns over large areas [
14]. Consequently, the field of biofabrication urgently requires a new manufacturing platform that is cost-effective while maintaining high precision and reproducibility, offering rapid production speed, and providing the flexibility to create diverse structures. In response to this technological demand, this study proposes a novel concept of a mask projection photolithography system that actively controls optical magnification by combining a physical mask with interchangeable objective lenses. The key advantage of this technology lies in its ability to optically control the size of the final structure derived from the fixed pattern of a single mask. This allows for the fabrication of various micro-scale structures without an expensive DMD, maximizing cost-effectiveness, while retaining the high-throughput advantage of projection methods that expose the entire area at once. The ultimate goal of this study is to rapidly fabricate PEGDA hydrogel microstructures with periodic porous networks using this innovative system and to validate its performance, thereby presenting its potential as a versatile platform for advanced biofabrication.
2. Experimental Methods
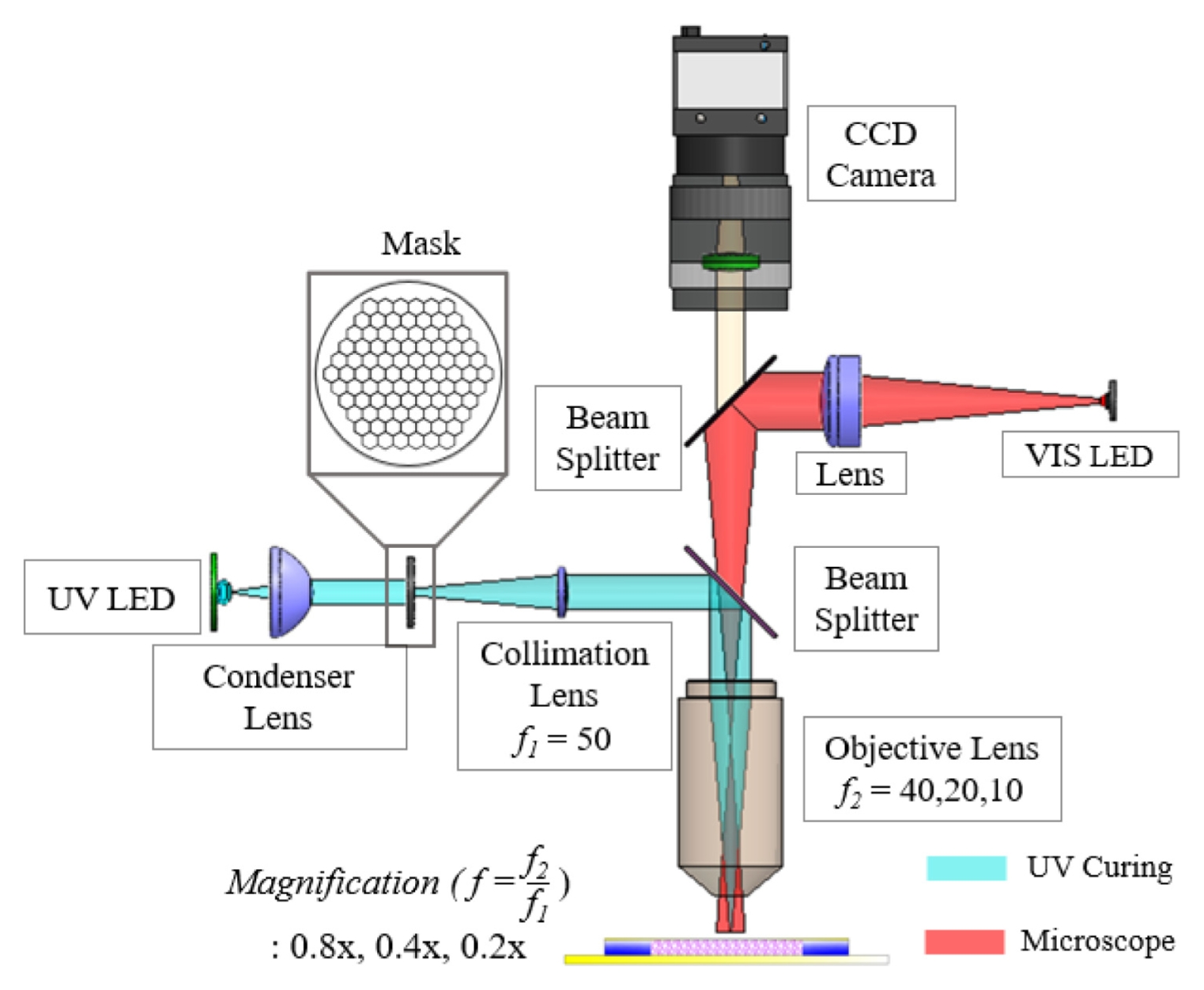
Fig. 1 illustrates the optical system used in this study. The system was designed based on an infinity-corrected platform that integrates a photopolymerization module and a real-time observation module.
An infinity-corrected configuration provides the advantage of modularity, allowing additional components such as a relay lens and a beam splitter to be inserted into the parallel optical path without introducing significant distortion [
15,
16]. The photopolymerization module employed a UV LED (M365L3, Thorlabs; central wavelength 365 nm, bandwidth 9 nm) as the excitation source, which matches the absorption spectrum of the photoinitiator Irgacure 2959 [
17]. The divergent light emitted from the LED was collected and transformed into a near-collimated uniform beam by a condenser lens, providing homogeneous illumination at the mask plane [
18].
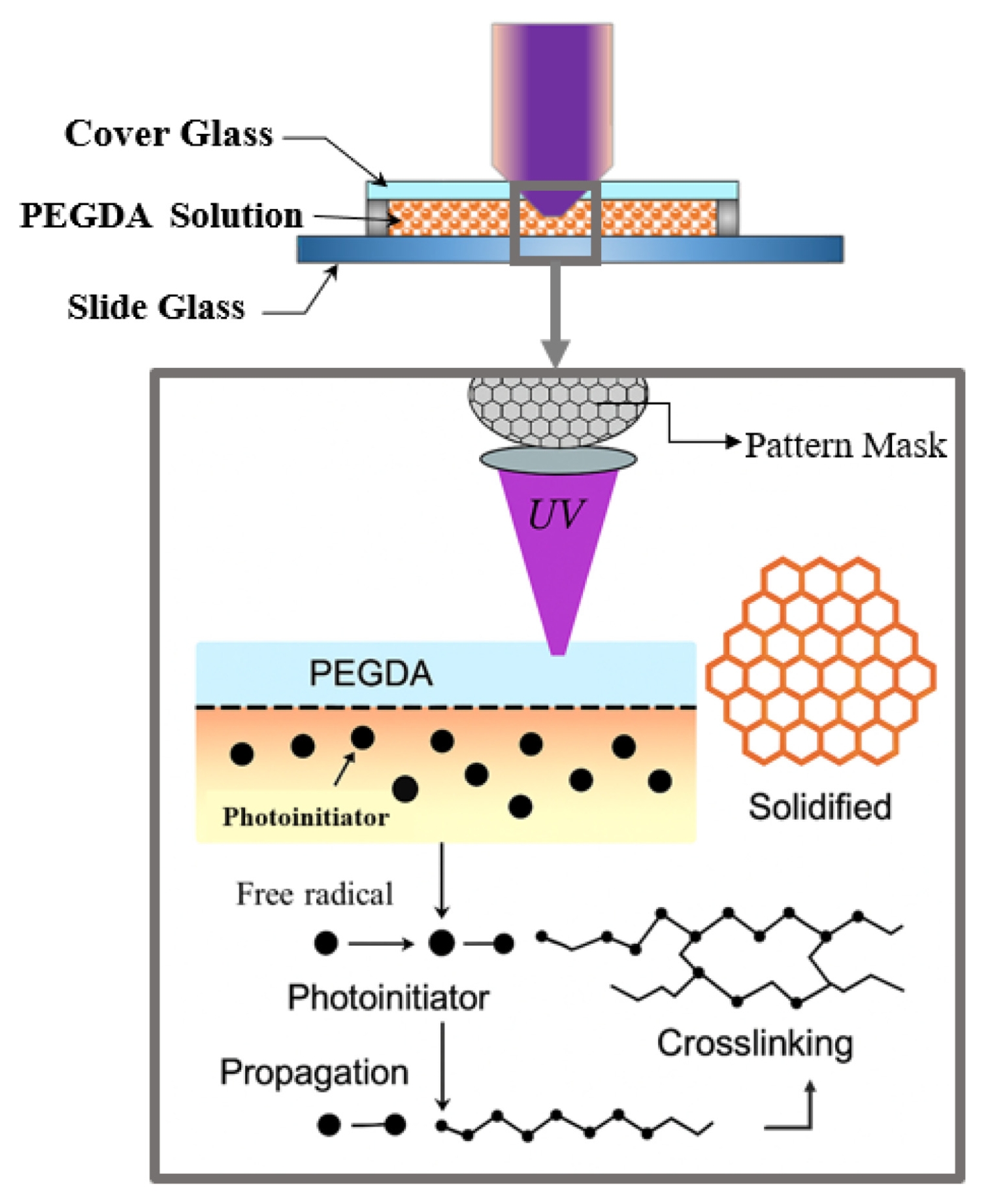
Fig. 2 shows the honeycomb-patterned physical mask placed at this mask plane, together with a schematic illustration of the PEGDA photopolymerization process. The patterned light transmitted through the mask was relayed by a 50-mm focal-length relay/collimation lens (Thorlabs; UV AR-coated), which transferred the mask image toward the entrance pupil of the objective lens. The interchangeable infinity-corrected objective lenses (5×, 10×, 20×) then determined the final optical reduction magnifications (0.8×, 0.4×, 0.2×, respectively) and projected the pattern onto the hydrogel precursor surface, enabling rapid single-shot fabrication of 2D microstructures over a large area. To clarify the focusing tolerance and reproducibility of the projection module, CFI LU Plan Fluor BD objective lenses (Nikon; 5×, 10×, 20×) were used, having numerical apertures (NA) of 0.15, 0.30, and 0.45 and working distances of 18, 15, and 4.5 mm, respectively. The corresponding depth of focus (DOF) was estimated using the Rayleigh approximation DOF ≈
λ/NA2 at 365 nm, yielding approximately 16.2 (5×), 4.1 (10×), and 1.8 μm (20×). It is worth noting that the Rayleigh-estimated DOF represents the theoretical diffraction-limited focusing tolerance, while the manufacturer-reported DOF corresponds to a practical contrast-based focusing criterion; however, both consistently indicate that higher NA results in a shallower focusing range. These quantitative optical specifications ensure that the projected pattern remains well-focused during exposure, supporting reliable pore size reproduction across the magnification conditions.
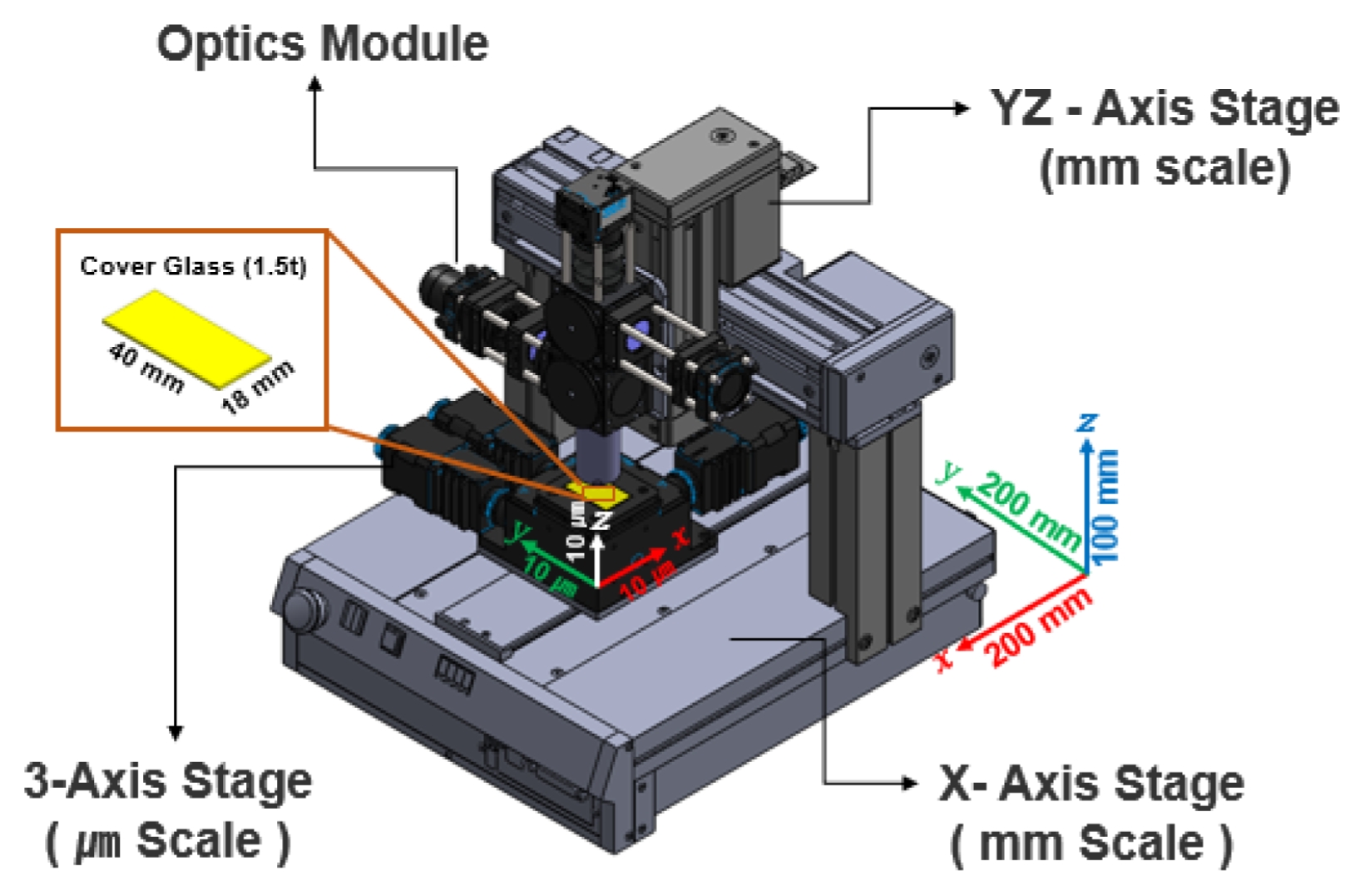
Figure 3 displays the precision 3-axis motorized stage integrated into the system. It provides micrometer-level repeat precision and was used for accurate focus control (Z-axis) and sample positioning (X-Y axis).
Poly(ethylene glycol) diacrylate (PEGDA, Mn = 700), known for its excellent biocompatibility, was used as the photocurable polymer. The PEGDA monomer was dissolved in ethanol at a 1:4 (v/v) ratio to reduce its viscosity and improve handling. Subsequently, the photoinitiator Irgacure 2959 was added to a concentration of 0.5 wt% of the total solution and completely dissolved using a magnetic stirrer in a dark environment to prepare the precursor solution. The principle of this process relies on the decomposition of Irgacure 2959 upon UV exposure to generate free radicals, which initiate chain polymerization of the terminal acrylate groups of PEGDA, forming a 3D crosslinked hydrogel network. The micropatterning process began by dispensing a specific amount of the PEGDA precursor solution onto a glass slide. The Z-axis stage was then adjusted to focus the objective lens on the solution surface. With the honeycomb mask in place, the UV LED was turned on to expose the pattern for a duration optimized for each objective lens’s magnification and light intensity. Specifically, the exposure times were 10 seconds for the 5× objective lens (0.8× reduction), 6 seconds for the 10× objective lens (0.4× reduction), and 3 seconds for the 20× objective lens (0.2× reduction). After patterning, the unreacted precursor solution was removed by washing the sample several times with ethanol. The samples were then dried, and the geometry and dimensions of the fabricated hydrogel structures were observed and measured using an optical microscope.
3. Results and Discussion
The optical performance of the system was validated prior to the experiments through optical simulations (Zemax OpticStudio).
Fig. 4 presents the spot diagram simulation results for the imaging optical module at each reduction magnification. From these results, it can be confirmed that the geometric radius of the spot formed on the image plane systematically decreases as the optical reduction magnification increases from 0.8× to 0.2×. A spot diagram illustrates how finely light is focused into a point on the image plane after passing through an optical system; a smaller and denser spot indicates higher resolution. The simulation results demonstrate that the designed optical system has sufficient performance to theoretically form high-resolution patterns at each magnification, providing a crucial basis for validating the feasibility of the actual system fabrication.
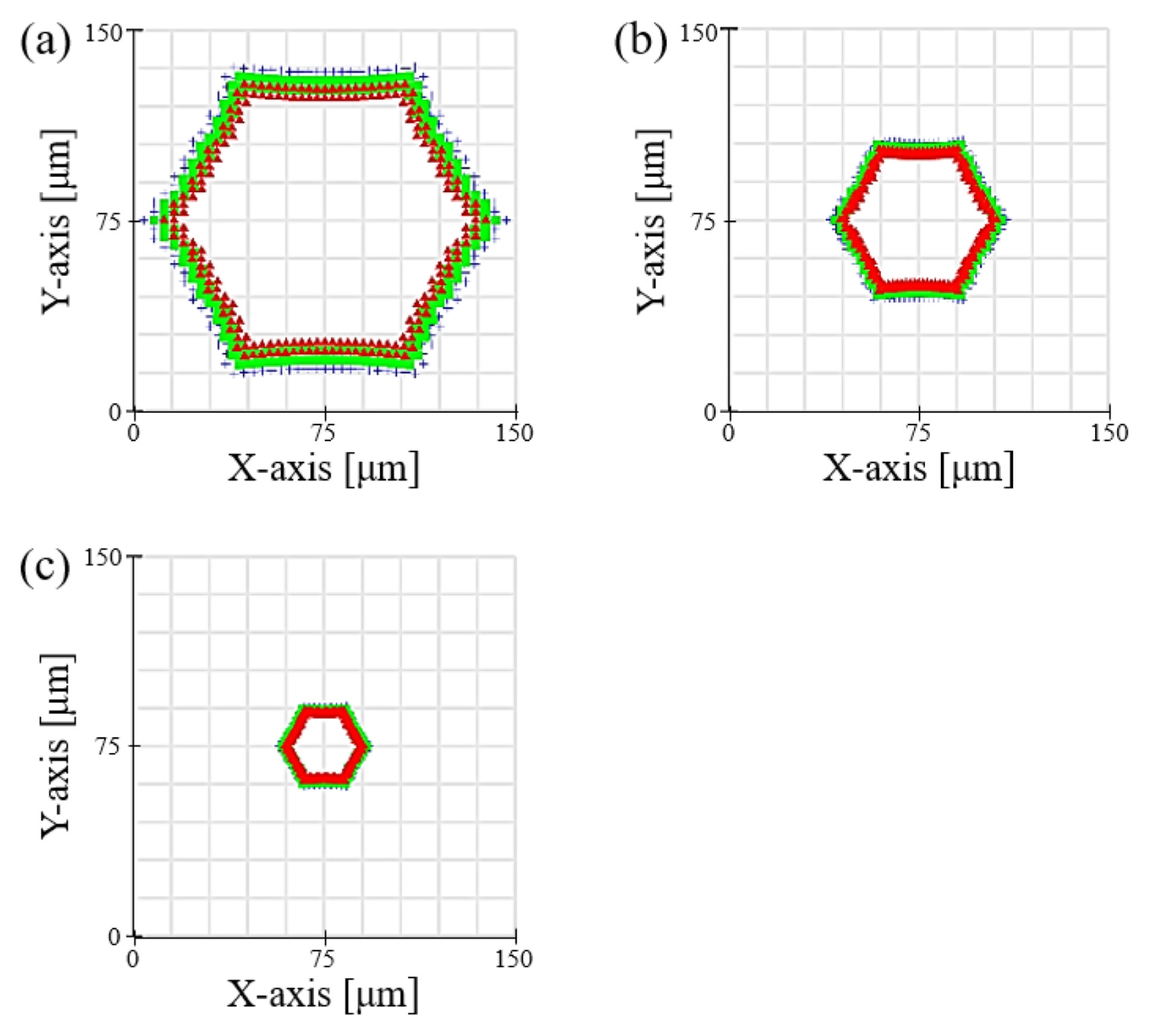
Fig. 5 shows the actual patterning results from the system fabricated based on the simulation results. It is evident that the honeycomb pattern was transferred into the hydrogel with very high fidelity under all conditions. The fabricated structures formed a uniform and perfectly interconnected porous network, which is an essential characteristic for cell migration and nutrient supply within the scaffold.
Table 1 summarizes the average pore diameters measured at each magnification. To ensure reproducible pore size evaluation, the optical microscope images in
Figs. 5(a)-
5(c) were analyzed using an image-based measurement procedure. The pixel-to-micrometer conversion factor was calibrated using the scale bar in each image, and the pore boundaries were segmented to extract the pore area
A. Although the pore geometry follows a hexagonal lattice, the pore size was represented using the equivalent circular diameter
Deq=2A/π, which is a standard descriptor for comparing non-circular pore structures based on area. In the overlaid images, the red crosses indicate the centroid of each pore, while the green circles represent uniform-radius overlays corresponding to the mean
Deq for each magnification condition. This confirmed that more than 95% of the pores across the patterned region were consistently detected, demonstrating high uniformity in pore size across the scaffold. Results of 100.5 ± 4.2 μm with the 5× objective lens (0.8× reduction), 51.2 ± 2.8 μm with the 10× objective lens (0.4× reduction), and 25.8 ± 1.9 μm with the 20× objective lens (0.2× reduction) were obtained. The very small standard deviation in the measured values experimentally demonstrates the high precision and reproducibility of this system. Notably, these results were achieved with a rapid, single exposure process lasting only 3 to 10 seconds, which clearly shows the high-throughput capability of the system. This phenomenon of pore size control can be explained by the optical property that the Numerical Aperture (NA) increases as the magnification of the objective lens increases. According to the Rayleigh criterion, the resolution of an optical system is inversely proportional to the NA; thus, a 20× objective lens with a high NA can create finer pores. The key significance of this research is that it presents a cost-effective and practical method for precisely controlling the feature size of the final structure in the range of tens of micrometers simply by changing the objective lens, without the need for expensive DMDs or complex laser scanning equipment. The ability of this system to precisely control pore sizes in the range of 25 to 100 μm is of great importance from a tissue engineering perspective. The 'optimal' pore size for a scaffold varies depending on the type of tissue to be regenerated. For instance, in cartilage regeneration (chondrogenesis), smaller pores of 25-50 μm are advantageous for promoting initial cell attachment and enhancing cell-to-cell signaling by providing a high specific surface area. In contrast, for bone regeneration (osteogenesis), larger pores of 100 μm or more are essential for securing space for vascularization and osteocyte infiltration. Achieving a uniform and interconnected porous network with precise pore size control (ranging from 25 to 100 μm) establishes the morphological foundation for the scaffold's structural stability. Moreover, these structural characteristics play a critical role in mimicking the extracellular matrix (ECM) microenvironment of specific target tissues, such as the aforementioned cartilage or bone. Therefore, the wide range of pore size control offered by this system provides a powerful experimental platform not just for creating structures, but for systematically studying and optimizing the scaffold microenvironment to match specific cell types and regeneration goals (e.g., cartilage vs. bone), which constitutes its academic significance. The potential biological applicability of this system could be further expanded if future studies incorporate the optimization of mechanical properties, such as compressive modulus, by adjusting the PEGDA concentration.
4. Conclusion
In this study to overcome the technical limitations of scaffold fabrication for tissue engineering, a new mask projection photolithography system capable of optical magnification control was successfully designed, fabricated, and validated. The developed system was demonstrated to provide an excellent combination of high throughput, high resolution, and process flexibility. The main achievements are as follows. First, the ability to rapidly fabricate porous hydrogel scaffolds with precisely controlled pore sizes within the biologically significant range of 25-100 μm in a matter of seconds using a projection mode was confirmed. Second, it was shown that the microdimensions of the final structure could be effectively controlled simply by changing the objective lens, enabling the fabrication of customized scaffolds for various tissue engineering needs. In conclusion, this research expands the horizons of biofabrication technology through an efficient approach of mask projection patterning and lays a solid foundation for its development as a core tool for understanding and reconstructing complex biological systems in the future.
FOOTNOTES
-
ACKNOWLEDGEMENT
This work was supported by the Korea Institute of Energy Technology Evaluation and Planning (KETEP) and the Ministry of Trade, Industry & Energy (MOTIE) of the Republic of Korea (No. RS-2024-00400653).
Fig. 1Schematic diagram of the mask projection photolithography system with variable optical magnification, integrating a UV curing module (cyan) and a microscopic observation module (red)
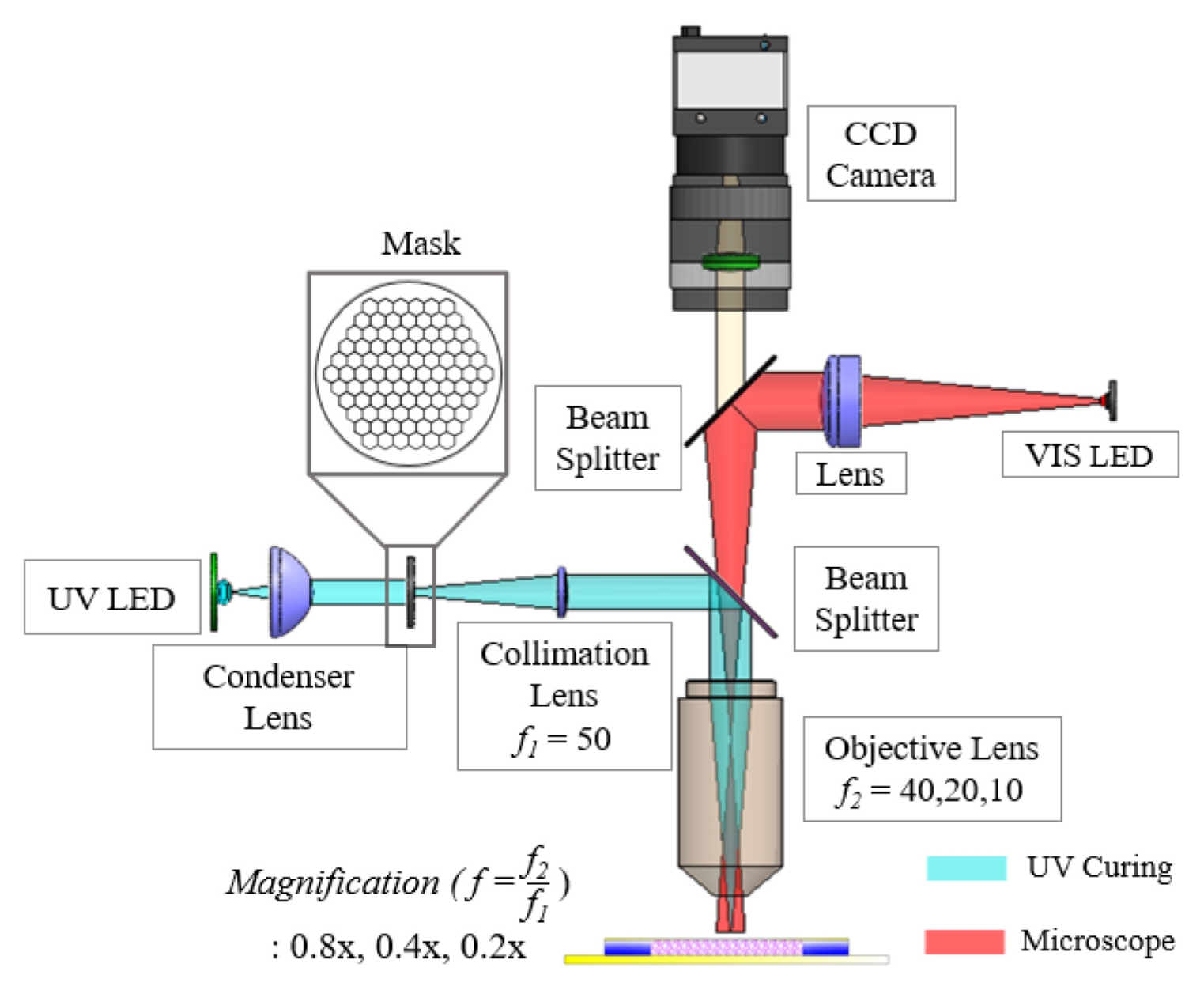
Fig. 2The photopolymerization mechanism of PEGDA, including photoinitiation, radical propagation, and final crosslinking under patterned UV light
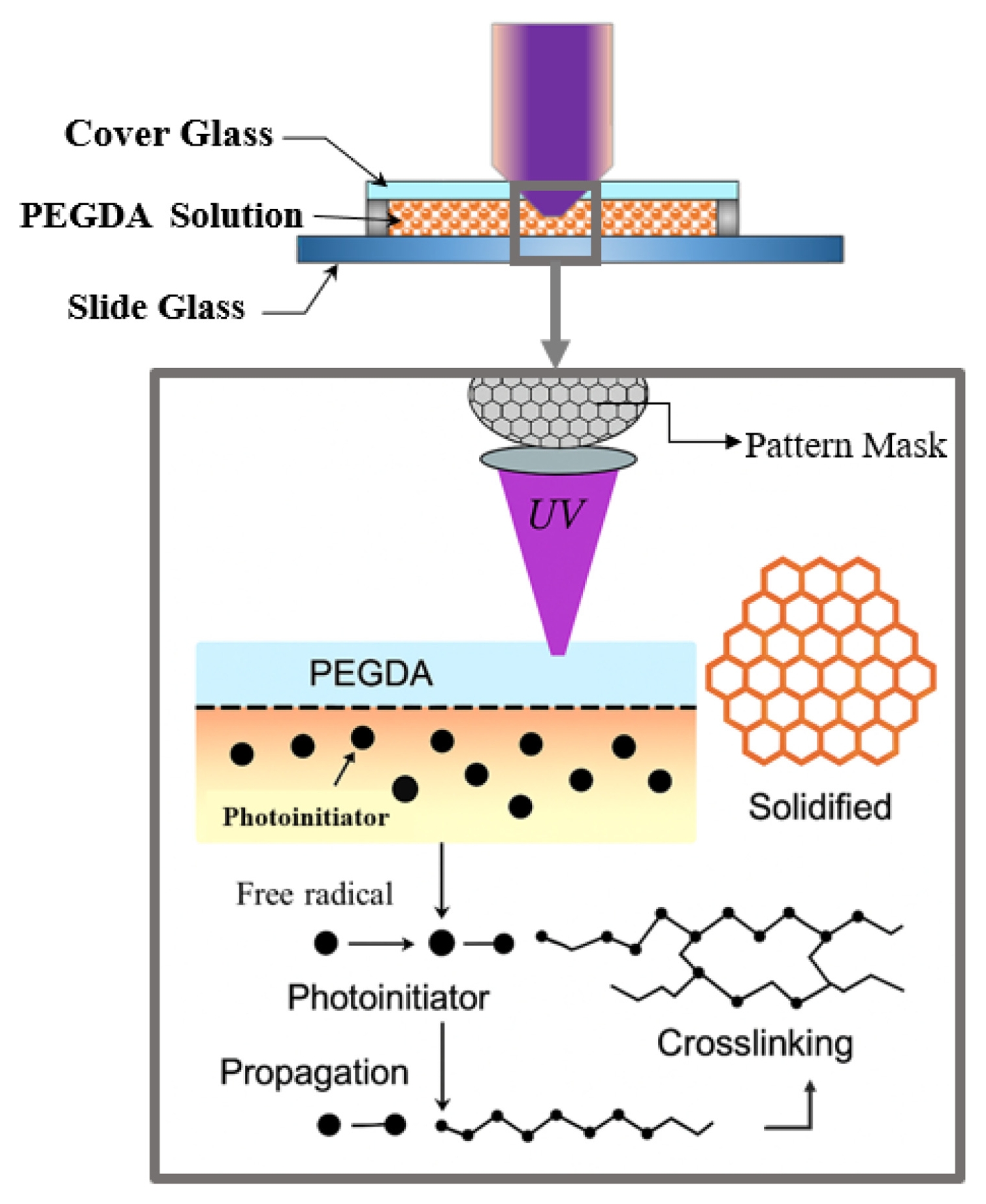
Fig. 3Configuration of the motorized stage system for sample positioning and optical focusing
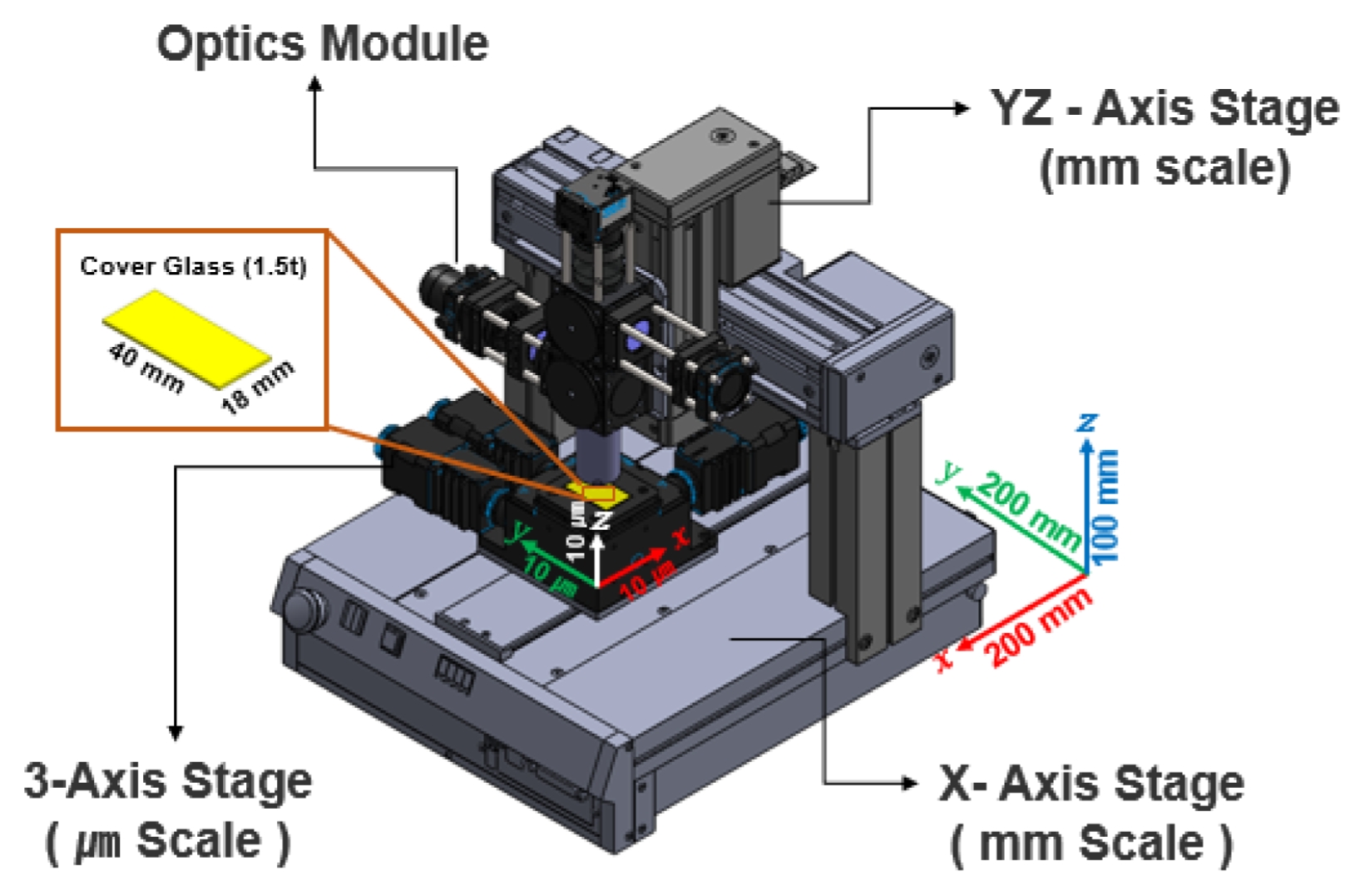
Fig. 4Spot diagram simulations of the imaging optical module showing chromatic performance at different reduction magnifications: (a) 0.8×, (b) 0.4×, and (c) 0.2×. The wavelengths simulated are 370 (red), 365 (green), and 360 (blue)
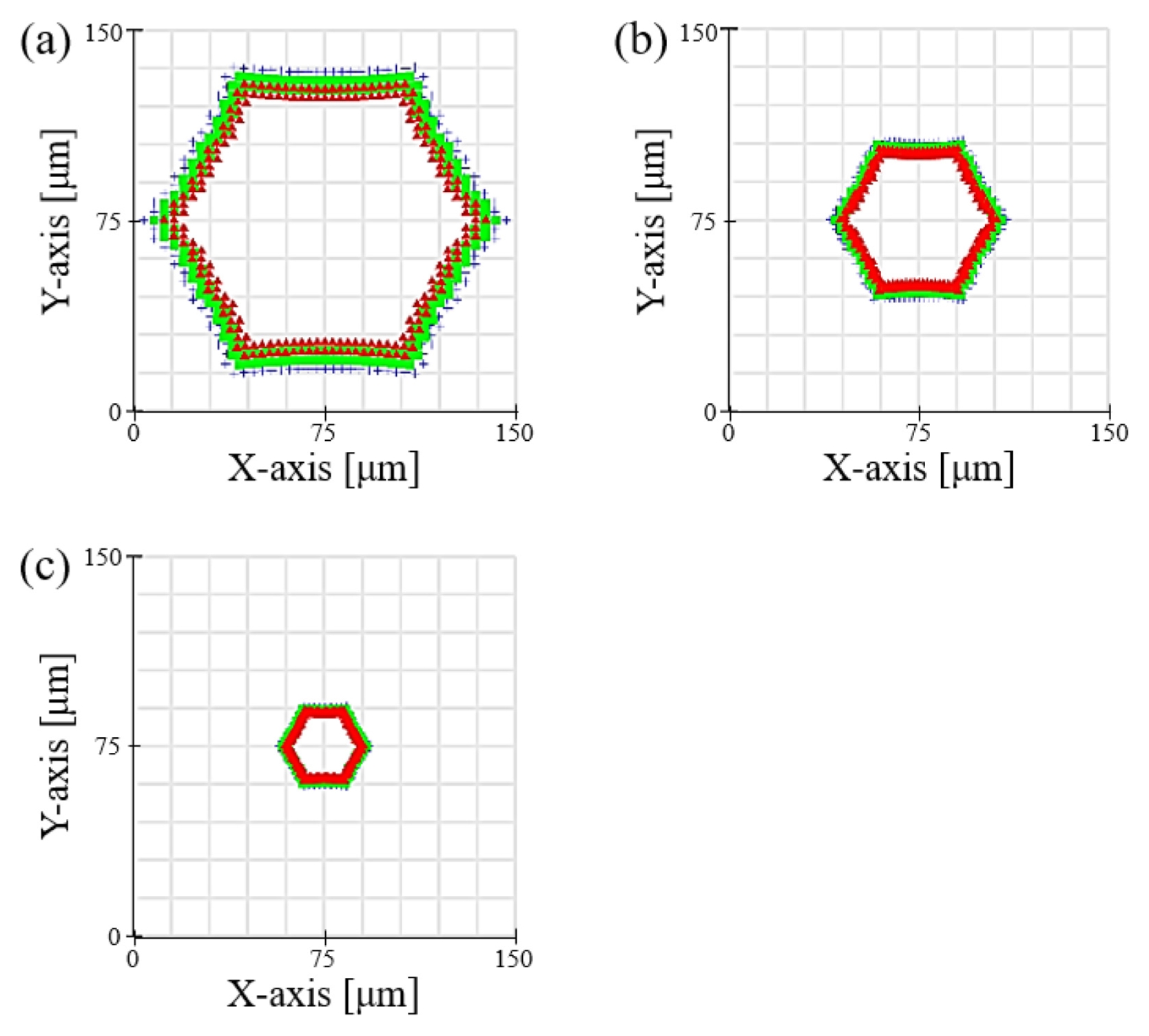
Fig. 5Optical microscope images of hexagonal hydrogel scaffolds fabricated at reduction magnifications of (a) 0.8×, (b) 0.4×, and (c) 0.2×

Table 1Experimental parameters and resulting pore diameters for fabricated hydrogel scaffolds
Table 1
|
Mode |
Objective lens |
Optical reduction mag. |
Exposure time [s] |
Pore dia. [μm] |
|
Projected mask |
5× |
0.8× |
10 |
100.5 ± 4.2 |
|
10× |
0.4× |
6 |
51.2 ± 2.8 |
|
20× |
0.2× |
3 |
25.8 ± 1.9 |
REFERENCES
- 1. Langer, R., Vacanti, J. P., (1993), Tissue engineering, Science, 260(5110), 920-926.
- 2. Hutmacher, D. W., (2000), Scaffolds in tissue engineering bone and cartilage, Biomaterials, 21(24), 2529-2543.
- 3. Tibbitt, M. W., Anseth, K. S., (2009), Hydrogels as extracellular matrix mimics for 3D cell culture, Biotechnology and Bioengineering, 103(4), 655-663.
- 4. Hollister, S. J., (2005), Porous scaffold design for tissue engineering, Nature Materials, 4(7), 518-524.
- 5. O’Brien, F. J., (2011), Biomaterials & scaffolds for tissue engineering, Materials Today, 14(3), 88-95.
- 6. Place, E. S., George, J. H., Williams, C. K., Stevens, M. M., (2009), Synthetic polymer scaffolds for tissue engineering, Chemical Society Reviews, 38(4), 1139-1151.
- 7. Hutmacher, D. W., Schantz, J. T., Lam, C. X., Tan, K. C., Lim, T. C., (2007), State of the art and future directions of scaffold-based bone engineering from a biomaterials perspective, Journal of Tissue Engineering and Regenerative Medicine, 1(4), 245-260.
- 8. Li, W. J., Laurencin, C. T., Caterson, E. J., Tuan, R. S., Ko, F. K., (2002), Electrospun nanofibrous structure: a novel scaffold for tissue engineering, Journal of Biomedical Materials Research, 60(4), 613-621.
- 9. Li, D., Xia, Y., (2004), Electrospinning of nanofibers: reinventing the wheel?, Advanced Materials, 16(14), 1151-1170.
- 10. Murphy, S. V., Atala, A., (2014), 3D bioprinting of tissues and organs, Nature Biotechnology, 32(8), 773-785.
- 11. Melchels, F. P. W., Domingos, M. A. N., Klein, T. J., Malda, J., Bartolo, P. J., Hutmacher, D. W., (2012), Additive manufacturing of tissues and organs, Progress in Polymer Science, 37(8), 1079-1104.
- 12. Derby, B., (2012), Printing and prototyping of tissues and scaffolds, Science, 338(6109), 921-926.
- 13. Malda, J., Visser, J., Melchels, F. P., Jüngst, T., Hennink, W. E., Dhert, W. J. A., Groll, J., Hutmacher, D. W., (2013), 25th anniversary article: Engineering hydrogels for biofabrication, Advanced Materials, 25(36), 5011-5028.
- 14. Cui, X., Breitenkamp, K., Finn, M. G., Lotz, M., D’Lima, D. D., (2012), Direct human cartilage repair using three-dimensional bioprinting technology, Tissue Engineering Part A, 18(11–12), 1304-1312.
- 15. Wang, C., Chen, X., Hong, J., Meng, L., Cheng, W., Zhu, X., Li, P., (2020), Extendable, large-field multi-modal optical imaging system for measuring tissue hemodynamics, Biomedical Optics Express, 11(5), 2339-2351.
- 16. Greb, C., Ferraro, P., (2016), Infinity optical systems, Optik & Photonik, 11(1), 34-37.
- 17. Bryant, S. J., Nuttelman, C. R., Anseth, K. S., (2000), Cytocompatibility of uv and visible light photoinitiating systems on cultured NIH/3T3 fibroblasts in vitro, Journal of Biomaterials Science, Polymer Edition, 11(5), 439-457.
- 18. Ostmann, S., Kähler, C. J., (2022), A simple projection photolithography method for low-cost rapid prototyping of microfluidic chips, Microfluidics and Nanofluidics, 26(3), 24.
Biography
- Sang Seon Lee
Ph.D. candidate in the Energy & Nano Technology Group, Korea Institute of Industrial Technology.
- Jae Cheol Park
Ph.D. in the Purpose Built Mobility Group, Korea Institute of Industrial Technology.